Influence of prone positioning on gross motor development in children with Spastic Diplegic Cerebral Palsy
Amir N. Wadee1*, Sara Samir Saad El-Dien2, Ahmed M. Elshinnawy3
1 Physical Therapy Department for Basic Sciences, Cairo University, Giza, Egypt. 2 Physical Therapy Department of Pediatric and its Surgery, Faculty of Physical Therapy, Modern University for Technology and Information, Cairo, Egypt. 3 Physical Therapy Department of Neuromuscular Disorder and its Surgery, Faculty of Physical Therapy, Modern University for Technology and Information, Cairo, Egypt.
ABSTRACT
Background: Cerebral palsy (CP), a heterogeneous disorder of gross motor development, is one of the most important causes of disability influencing children. Objective: To detect the influence of prone positioning on gross motor development in children with spastic diplegic cerebral palsy. Methods: Forty-two spastic diplegic CP children of both genders participated in this study. They were randomly chosen from the Medical Centre of Faculty of Physical Therapy, Modern University for Technology and Information, Cairo, Egypt. Their ages ranged from 12 to 18 months, with partial control of head and trunk according to gross motor function measure (GMFM), and mild spasticity (grade 1, 1+ according to Modified Ashworth scale(. Children were randomly assigned into control group, received selected physical therapy program based on the neurodevelopmental principles for 60 minutes, and study group received the selected physical therapy program in addition to a designed prone and quadruped exercise for 30 minutes. Treatment procedures conducted three times/week for three successive months. Results: The in-between comparison of GMFM showed no significant differences in the pre-measures while there were significant differences in the post-measures (t= 1.322, P= 0.194, t= 6.397, and P= 0.000, respectively). Conclusion: The results of this study revealed that prone position training program was effective in improving the gross motor skills in children with spastic diplegia.
Keywords: Prone Positioning, Gross Motor Development, Spastic Diplegic Cerebral Palsy.
Introduction
Cerebral Palsy (CP) is a congenital non-progressive neurodevelopmental disorder that influences muscle tone, movement, balance, and motor control. The cause of CP is not always known but is typically the result of a prenatal or perinatal problem when the fetus’ brain does not develop normally or during the first five years of life as a result of trauma to the brain [1]. Insufficient monitoring perinatal period, increase unreported fetus health issues [2]. Besides, maternal health affects the health of babies [3] and even may cause perinatal mortality [4].
The clinical signs relay upon the size, degree, and area of the lesion. Despite the lesion is non-progressive, the signs and symptoms are variable. Improper treatment increases the risk for critical issues such as muscle and soft tissue contractures as well as bony deformities [5].
Spastic diplegic CP is a typical type of CP, characterized by more affection of the lower limbs, although the upper limbs are not spared. It is strongly related to prematurity. The periventricular germinal matrix is commonly affected by hemorrhage and hypoxic-ischemic insults [6].
Normal infants were more advanced in their early motor development if they slept in the prone position, but were less well developed if they slept in the supine position. This has been confirmed with recent research by comparing the motor skills of prone and supine sleepers and determined that at age of six months, prone sleepers demonstrated significantly higher motor scores than supine sleepers, whereas 22% of supine sleepers exhibited gross motor delay [7].
Infants who are positioned prone utilize muscles that are essential for head control, pulling up, reaching, crawling, and other developmental milestones. Tummy time gives the infant opportunities to strengthen the muscles that are essential for these milestones, such as arm, shoulder girdle, and neck muscles [8].
To our knowledge, there was a lack of researches that studied the influence of prone positioning on the gross motor development of children with CP. So, this study aimed to study the impact of prone positioning on gross motor development in children with spastic diplegic cerebral palsy. It was hypothesized that there was no significant difference in gross motor function measure (GMFM) between children with spastic diplegic CP who received prone positioning and those who did not.
Subjects, Materials, and Methods
The study design was a randomized control trial, which was conducted between December 2018 to May 2019.
Ethical considerations
The study was approved by the ethical committee of the Faculty of Physical Therapy, Cairo University, Egypt by P.T.REC/012/002375. Two copies of the written consent form were signed by the children’s parents for participation and publications of results.
Participants
Forty-two spastic diplegic CP children of both genders participated in this study. They were randomly selected based on the following inclusion criteria: their age ranged from 12 to 18 months, they had partial control of head and trunk according to GMFM, and mild spasticity (grade 1, 1+ according to Modified Ashworth scale). Exclusion criteria included: children with visual or auditory problems, epilepsy, and those with fixed deformities in upper limbs, lower limbs or trunk.
Forty-nine children with spastic diplegia were initially screened and assessed to determine age, diagnosis, inclusion, and exclusion criteria. From the screened children, only forty-five children fulfilled the inclusion criteria, one parent refused to participate in the study while the other two children didn’t follow the designed program and were excluded from the study. Thus, of the original pool, forty-two children with spastic diplegia were included in this study.
Children were randomly selected, using the roll of a dice, from the medical Centre of Faculty of Physical Therapy, Modern University for Technology and Information, Cairo, Egypt. Then, the random assignment of the children to one of the two groups (study and control groups) was done by using sealed envelopes. The randomization process was carried out by a registration clerk who was not involved in any part of the study (figure 1).
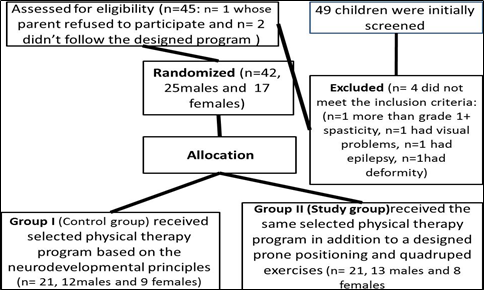
Figure 1: The dropout and allocation.
Children in the control group received a selected physical therapy program based on the neurodevelopmental principles for 60 minutes. While those in the study group received the same selected physical therapy program in addition to a designed prone positioning and quadruped exercises for 30 minutes. The treatment procedures were performed three times/week for three successive months for both groups.
Materials and procedures
A. Evaluative Instruments
1) Gross Motor Function Measure-88
The motor function of children with CP is usually evaluated utilizing the Gross Motor Function Measure (GMFM-88). It is an evaluative, standardized, criterion-referenced observational instrument that was developed to determine changes in gross motor function over time in individuals with CP. This measure has been considered for its reliability (intra-rater, test-retest, and interrater) and validity [9].
The GMFM-88 was a criterion-referenced measure utilized to evaluate motor function in children with CP.15. The GMFM-88 evaluated 66 different items under five different dimensions labeled A to E. These dimensions included: lying and rolling (Dimension A), sitting (Dimension B), kneeling and crawling (Dimension C), standing (Dimension D), and walking, running and jumping (Dimension E). Each item was scored on a 0-3 point scale with a lower score indicating greater impairment in gross motor function [10].
2) Modified Ashworth scale
The Modified Ashworth scale was used to assess the degree of spasticity for all children in both groups before starting the treatment. The Modified Ashworth scale MAS used a 6 point scale (0, 1, 1+, 2, 3, and 4) with "0" signifying no tone and “4” signifying a rigid limb [11].
B. Treatment Instruments
Physical therapy tools were used for conducting the selected physical therapy program for both groups including different sizes of mats, wedges, rolls, blocks, medical balls, balance, and rocker boards. Besides, special occupational therapy tools and visual tools were used during the prone and quadruped exercises to motivate the child.
Interventions
The selected physical therapy program
Facilitation of motor development: Children in both control and study groups received selected physical therapy programs including facilitation of head and trunk control, segmental rolling, and sitting milestones as well as facilitation of postural reactions, equilibrium, and protective reactions.
Flexibility exercises: These exercises were conducted to muscles and soft tissues liable to be tightened especially the Achilles tendon, hamstrings, hip flexors and adductors in the lower limbs and the shoulder internal rotators, elbow and wrist flexor, pronators of the upper limbs, to maintain the length and elastic recoil of the tight muscles. It was applied for 20 seconds and 20 seconds rest within the limit of pain with five repetitions [12]
Designed a program for study group: The program was applied in a quiet room. The program included:
A. From Prone positioning on the wedge: with the child’s head and upper trunk outside the wedge:
- The child encouraged to reach the object at a different height.
- The child encouraged to track bright objects at different heights and directions.
- Facilitation of head and trunk extension using different kinds of tactile stimulation paraspinal.
B. From quadruped position: With hand weight bearing on one upper limb and trying to reach object above the head with the other hand, alternatively [13].
Statistical Analysis:
All statistical calculations were performed utilizing the computer program IBM SPSS version 22 (IBM Corporation, USA). The sample size calculations were performed using the G*Power software (version 3.0.10). The gross motor function measure (GMFM) score was selected as the primary outcome measure. The effect size of GMFM was estimated to be medium (0.25). A generated sample size of at least 17 patients per group would be required. Allowing for a 20% dropout rate, it was necessary to reach a total sample level of a minimum of 42 patients. Test of homogeneity (Levene's test) revealed that all data were homogenous. Test of normality (Shapiro-Wilk test) was used before applying statistical analysis, and it showed that data were normally distributed, so parametric test was used (paired t-test within groups and independent t-test in-between groups). P-values less than 0.05 were considered statistically significant.
Results:
- General demographic data:
The children' age was ranged from 12 to 18 months (15.2 + 2.30 M), height was ranged from 74 to 80 cm (76.8 + 2.27Cm) and weight was ranged from 9.5 to 12 Kg (10.7 + 0.92Kg). The demographic data showed no significant differences in-between control and study groups (t=0.156, P= 0.877, t= 0.158, P= 0.875, t= 0.393, and P= 0.697, respectively) (Table 1 and Figure 2).
|
Table 1: General demographic data. |
|||||
|
Groups |
Age (M) |
Gender |
Height (Cm) |
Weight (Kg) |
|
|
Males |
Females |
||||
|
Whole Sample |
15.2 + 2.30 |
25 |
17 |
76.87 + 2.27 |
10.7 + 0.92 |
|
Control |
15.27 + 2.43 |
12 |
9 |
76.87 + 2.39 |
10.77 + 0.98 |
|
Study |
15.13 + 2.23 |
13 |
8 |
76.73 + 2.22 |
10.63 + 0.77 |
|
t-value |
0.156 |
|
|
0.158 |
0.393 |
|
P-value |
0.877 |
|
|
0.875 |
0.697 |
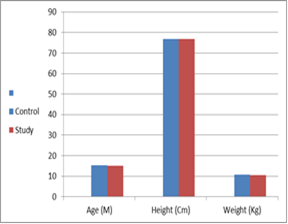
Figure 2: General demographic data.
- GMFM:
In the control group, the mean pre-value was 10.33 + 1.11 and the post-value was 11.71 + 0.644. This indicated significant differences within measures (t= 6.874, P= 0.000) with the percentage of improvement of 10.74%.
In the study group, the mean pre-value was 9.81 + 1.44 and the post-value became 13.62 + 1.2. This indicated significant differences within measures (t= 13.152, P= 0.000) with the percentage of improvement of 12.62%.
The in-between comparison between the control and study groups showed no significant differences in the pre-measures while significant differences of the post-measures (t= 1.322, P= 0.194, t= 6.397 and P= 0.000 respectively) (Table 2 and Figure 3).
|
Table 2: GMFM within and in-between groups. |
|||||
|
|
Pre- test |
Post- test |
% of Improvement |
t- value |
P-value |
|
Control |
10.33 + 1.11 |
11.71 + 0.644 |
10.74% |
6.874 |
0.000* |
|
Study |
9.81 + 1.44 |
13.62 + 1.2 |
12.62% |
13.152 |
0.000* |
|
t- value |
1.322 |
6.397 |
|
|
|
|
P-value |
0.194 |
0.000* |
|
|
|
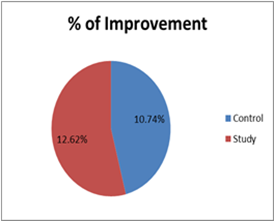
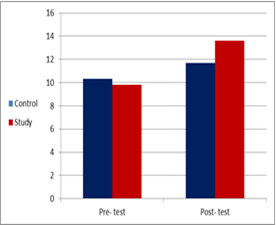
Figure 3: GMFM within and in-between groups.
Discussion:
Prone positioning is an important and critical part of the child's daily routine. It can be carried out at many different times throughout the day. It does not have to be limited to placing the infant on the floor but can be carried out while holding, carrying, and even dressing a baby which comes in agreement with Kuo et al. [14] who stated that parents should be informed that initial exposure to prone positioning can be implemented in a variety of ways, such as placing the baby on a caregiver’s chest when the caregiver is in a reclined position, placing baby on the caregiver’s lap, holding and carrying the infant in a prone position, and burping the baby in a prone position on the caregiver’s lap. So, in the study group, there were significant differences within measures (t= 13.152, P= 0.000).
The findings of the current study agree with Aliberti et al. [15] who found a significant difference in age for the attainment of many early motor milestones including rolling, sitting, creeping, crawling, and pulling to stand with the prone sleepers acquiring the milestones earlier than those sleep in supine. Similarly, Salls et al. [16] stated that regular prone positioning allows infants to develop head control at an early stage of life. Neck muscles are strengthened as they attempt to lift their heads to look around. Chizawsky et al. [17] mentioned that there are significant differences in the active movements of the arms and the pushing-up on the arms between the prone and non-prone infants.
Moreover, a significant difference between both groups with higher improvement in the study group may be related to the designed prone position program which was based on sensory stimulation including tactile stimulation, proprioceptive stimulation through weight-bearing positions, and stroking on the child's back from a prone position to enhance the gross motor function. So, the percentage of improvement in the control group was 10.74% while it was 12.62% in the study group. This opinion was supported by the study of Jennings et al. [18] who revealed that at ages of six months and 18 months, the average Peabody Developmental Motor Scales (PDMS–2) locomotion scores of babies consistently positioned in the prone position is higher than non-prone positioned children.
Moreover, varying positions for the infants were believed to be important for their motor development. Therefore, the control group indicated significant differences within measures (t= 6.874, P= 0.000). However, the prone position was of particular importance in the development of head control and antigravity extension which comes in consistence with Heard a Harris [19] who mentioned that prone extension is essential for the development of stability in various weight-bearing positions such as prone-on-hands, on all fours, and sitting. Hence, the lack of experience in the prone position may have a detrimental effect on the motor development of the infants.
Besides, placing the child in a prone position facilitates midline hand skills which come in consistence with Humphreys et al. [20] who found that placing the infant in prone position will gain upper body strength. So, the infant will be able to lift its head and chest in a more functional position to explore the environment on its own. So, the in-between comparison showed significant differences in the post-measures (t= 6.397, and P= 0.000).
Therefore, higher improvement in the gross motor abilities in the study group comes in agreement with Kajita [21] who mentioned that prone positioning encourages use of upper body strength used in acquisition of many infant motor milestones. It might be that supine sleepers lag in milestone development in the first year because the upper body contributes less to daily movement than for predominantly prone positioned infants. So, the previously stated hypothesis of no significant difference in gross motor function measure (GMFM) between children with spastic diplegic CP who received prone positioning and were not rejected.
Prone position not only affects the child's gross motor skills but also affects the child's social and intellectual properties. This was supported by the results of Gough [22] who found that prone sleepers had higher scores in gross motor, social skills, and overall development.
The improvements in overall gross motor performances of the participated children as head control, pulling up, reaching, crawling, and other developmental milestones in the current study contradict the findings of Higgins et al. [23] who stated that placing the child in prone position made them rolled over at an earlier age than non-prone sleepers. However, no significant difference was found in other milestones.
Conclusion
The results of this study revealed that the prone position training program was effective in improving the gross motor skills in children with spastic diplegia.
References
- Sankar C, Mundkur N. Cerebral palsy-definition, classification, etiology and early diagnosis. The Indian Journal of Pediatrics. 2005 Oct 1;72(10):865-8.
- El-Sayed SA, Shehab MM, Ahmady MM, Baraka A. Early Management of Newborn with Meconium Aspiration Syndrome Using Continuous Positive Airway Pressure as a Special Modality. International Journal of Pharmaceutical and Phytopharmacological Research. 2018 Feb 1;8(1):16-20.
- AlaeeKarahroudy F, Mahdavi Khanouki Z, Pour Arian Sh, Rezai-Zadeh A. The Correlation between Using Nasal CPAP Device and Neonatal Outcomes. Pharmacophore. 2018; 9(6): 22-25.
- Mehri M, Iravani M, Sayyah Bargard M, Haghighizadeh MH. Effectiveness of Cognitive Behavioral Therapy-based selfmanagement on depression in Pregnant Women: A Randomized Controlled Trial. J Biochem Tech. 2018; Special Issue (2): 95-100.
- Donnelly C, Parkes J, McDowell B, Duffy C. Lifestyle limitations of children and young people with severe cerebral palsy: a population study protocol. Journal of advanced nursing. 2008 Mar;61(5):557-69.
- Hoare B., Russo R. Botulinum toxin-A in children with cerebral palsy: Treatment of the upper limb following injection. In: Soderback, 1st (ed). International Handbook of Occupational Therapy Interventions, 2012.
- Dudek-Shriber L, Zelazny S. The effects of prone positioning on the quality and acquisition of developmental milestones in four-month-old infants. Pediatric Physical Therapy. 2007 Apr 1;19(1):48-55.
- Majnemer A, Barr RG. Association between sleep position and early motor development. The Journal of pediatrics. 2006 Nov 1;149(5):623-9.
- Lundkvist Josenby A, Jarnlo GB, Gummesson C, Nordmark E. Longitudinal construct validity of the GMFM-88 total score and goal total score and the GMFM-66 score in a 5-year follow-up study. Physical therapy. 2009 Apr 1;89(4):342-50.
- Russell DJ, Avery LM, Walter SD, Hanna SE, Bartlett DJ, Rosenbaum PL, Palisano RJ, Gorter JW. Development and validation of item sets to improve efficiency of administration of the 66‐item Gross Motor Function Measure in children with cerebral palsy. Developmental Medicine & Child Neurology. 2010 Feb;52(2):e48-54.
- Mutlu A, Livanelioglu A, Gunel MK. Reliability of Ashworth and Modified Ashworth scales in children with spastic cerebral palsy. BMC musculoskeletal disorders. 2008 Dec;9(1):44.
- Shamsoddini A. Comparison between the effect of neurodevelopmental treatment and sensory integration therapy on gross motor function in children with cerebral palsy. Iranian Journal of Child Neurology. 2010 Jun 1;4(1):31-8.
- Craig P, Dieppe P, Macintyre S, Michie S, Nazareth I, Petticrew M. Developing and evaluating complex interventions: the new Medical Research Council guidance. Bmj. 2008 Sep 29;337:a1655.
- Kuo YL, Liao HF, Chen PC, Hsieh WS, Hwang AW. The influence of wakeful prone positioning on motor development during the early life. Journal of Developmental & Behavioral Pediatrics. 2008 Oct 1;29(5):367-76.
- Aliberti F, Pittore L, Ruggiero C, Cinalli G, Maggi G. The treatment of the positional plagiocephaly with a new thermoplastic orthotic device. Child's nervous system. 2002 Jul 1;18(6-7):337-9.
- Salls JS, Silverman LN, Gatty CM. The relationship of infant sleep and play positioning to motor milestone achievement. American Journal of Occupational Therapy. 2002 Sep 1;56(5):577-80.
- Chizawsky, L.L.K, Scott-Findlay S. Tummy Time! Preventing Unwanted Effects of the “Back to Sleep” Campaign. AWHONN Lifelines. 2015; 9: 382-387.
- Jennings JT, Sarbaugh BG, Payne NS. Conveying the message about optimal infant positions. Physical & occupational therapy in pediatrics. 2005 Jan 1;25(3):3-
- Heard R. and Harris L.M. Experimental, quasi-experimental and correlational quantitative research designs. In: Minichiello V, Sullivan G, Greenwood K, Axford R, editors. Handbook of Research Methods for Nursing and Health Science. 2nd eds. Australia: Prentice-Hall. 2014; 124–150.
- Humphreys G, Mandy A, Pountney T. Posture and sleep in children with cerbral palsy: A case study. Association of Paediatric Chartered Physiotherapists. 2012 Jan 1;3:48-56.
- KAJITA. Pediatric Clinic (Japan). Yaki: improving Yaoki's body symmetry with the Leckey Sleepform System. 2010. http:// bit.ly/1yLdZY4.
- Gough M. Continuous postural management and the prevention of deformity in children with cerebral palsy: an appraisal. Developmental Medicine & Child Neurology. 2009 Feb;51(2):105-10.
- Higgins JPT, Altman DG, Sterne JAC. Chapter 8: Assessing the risk of bias in included studies. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0, 2014.
Contact SPER Publications
SPER Publications and
Solutions Pvt. Ltd.
HD - 236,
Near The Shri Ram Millenium School,
Sector 135,
Noida-Greater Noida Expressway,
Noida-201301 [Delhi-NCR] India
