Effect of drug treatment on serum Cartilage Oligomeric Matrix Protein levels in patients of Rheumatoid Arthritis and Knee Osteoarthritis in Bandung, Indonesia
Nyi Mekar Saptarini1*, Ginayanti Hadisoebroto2, Marlia Singgih Wibowo3, Tutus Gusdinar3
1 Department of Pharmaceutical Analysis and Medicinal Chemistry, Faculty of Pharmacy, Universitas Padjadjaran, Jatinangor 45363, Indonesia. 2 Department of Pharmacy, Faculty of Mathematics and Natural Sciences, Universitas Al Ghifari, Bandung 40293, Indonesia. 3 Department of Physicochemistry, School of Pharmacy, Institut Teknologi Bandung, Bandung 40243, Indonesia.
Correspondence: Nyi Mekar Saptarini; Department of Pharmaceutical Analysis and Medicinal Chemistry, Faculty of Pharmacy, Universitas Padjadjaran, Jatinangor 45363, Indonesia. Email: nyi.mekar @ unpad.ac.id.
|
ABSTRACT Joint illnesses such as rheumatoid arthritis (RA) and knee osteoarthritis (OA) are defined by the destruction of articular cartilage and changes of the underlying bone. Enhanced proteolysis of molecules bound to and exposed at the surface of collagen, such as cartilage oligomeric matrix protein (COMP) will make an imbalance in the turnover of matrix proteins. The objective of this investigation was to analyze the influence of drug treatment on COMP levels of treated RA and knee OA patients. The subjects were RA patients who visited the rheumatology clinic at one public hospital and knee OA patients who visited the Orthopedic clinics of a private hospital in Bandung, Indonesia. Prescribed drugs were collected utilizing interviews. COMP levels were measured by enzyme-linked immunosorbent assay. The COMP levels of RA patients, knee OA patients, and normal individuals were 873.16 ± 165.6, 911.43 ± 227.34, and 927.26 ± 90.38 ng/mL, respectively. There was no significant difference in the COMP level (p = 0.47) among the groups. Mostly, RA patients were treated with single or combined disease-modified antirheumatic drugs, while most of the knee OA patients were treated with glucosamine and chondroitin. Drug treatment maintained the serum COMP levels of RA and knee OA patients. Keywords: arthritis, ELISA, maintained COMP, serum, treated patient |
Introduction
Rheumatoid arthritis (RA) and osteoarthritis (OA) are joint diseases, which are indicated by articular cartilage damage and underlying bone alteration.[1] The turnover imbalance of matrix proteins leads to enhanced proteolysis of molecules bound to and exposed at the surface of collagen fibers, such as fibromodulin, decorin, and cartilage oligomeric matrix protein (COMP).[2-4] Early-stage diagnosis of cartilage degradation is important to minimize tissue rupture.[5]
COMP is a non-collagen extracellular matrix protein, which is mainly found in cartilage, that maintains the integrity of collagen fibrils.[6] The extracellular matrix, such as collagens I, II, and IX, are involved in interactions with the C-terminal domain of COMP.[7, 8] The C-terminal domain also binds collagen and catalyzes collagen fibril assembly.[7] COMP is cleaved and released from the cartilage into the synovial fluid in RA and OA,[6, 9, 10] thereby COMP is used as a cartilage turnover marker.[11] The aim of this study was to analyze the effect of drug treatment on COMP levels of treated RA and knee OA patients.
Subjects and Methods
Subjects
This investigation was verified by the Health Research Ethics Committee of Dr. Hasan Sadikin Public Hospital Bandung, No. LB.04.01/A05/EC/075III/2016. All subjects were recruited after being briefed and signing informed consent. The serum of RA patients were collected from the Rheumatology Clinic of Dr. Hasan Sadikin Public Hospital Bandung,[12] and the serum of OA patients were collected from the Orthopedy Clinic of a private hospital in Bandung, West Java, Indonesia. All patients were interviewed for the prescribed drug and disease duration.
Determination of serum COMP levels
The Human COMP/TSP5 ELISA Pair Set (SEK10173) from Sino Biological Inc. (USA) was used to determine the serum COMP levels. A 96-well-plate was coated with a monoclonal antibody specific for COMP/TSP5 as a capture antibody, then reacted with the analyte, i.e. standards and samples. COMP in analytes was bonded with the immobilized antibody. All wells were rinsed, then a biotinylated rabbit anti-COMP polyclonal antibody was added to generate an antibody-antigen-antibody pattern. The solution of horseradish peroxidase (HRP)-conjugated streptavidin, the substrate of tetramethylbenzidine, and sulfuric acid as a stop solution were loaded to produce a yellow color. Spectrophotometer at 450 nm was used to measure the absorbance levels, which indicated the COMP levels in serum.
Statistical analysis
R i386 3.2.0 software was used to analyze the correlation between disease duration and serum COMP levels in RA and OA patients.
Results and Discussion
Subject
Patients who met the inclusion criteria were 30 patients for RA and 18 patients for OA. RA is an autoimmune disease started in early adulthood, whereas OA is a degenerative disease is suffered in late adulthood until the elderly (Table 1). Patients lacked the knowledge or awareness of arthritis symptoms. This was observed from the interviews, where it was found that the patient visited the doctor after they felt unable to move. Heavy activities for more than 30 minutes causes severe pain that is treated with analgesics, such as diclofenac sodium or paracetamol.
|
Table 1: Anthropometric parameter comparison. |
|||||
|
|
Parameter |
|
|||
|
Group |
Age (years) |
Sex (M/F) |
|
||
|
|
Mean + SD |
Range |
|
||
|
RA patients |
44 ± 11 |
20 – 64 |
4/26 |
||
|
OA patients |
61 ± 7 |
47 - 75 |
2/16 |
||
|
Normal individuals |
41 ± 13 |
21 - 60 |
4/26 |
||
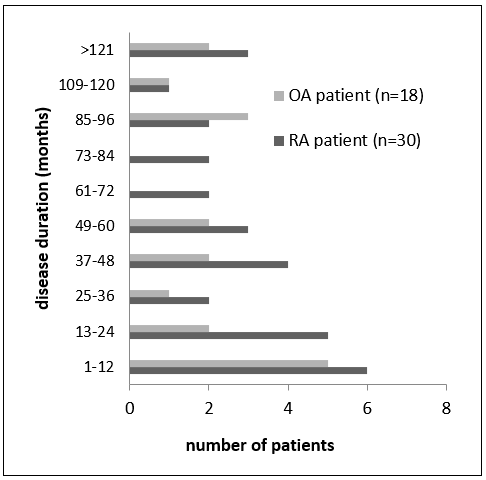
Figure 1. Distribution of patients to disease duration. The disease duration range was very wide with various conditions.
Health monitoring awareness due to joint pain was very low in patients under 50 years of age. This is because Indonesian people assume that RA and OA are elderly people’s diseases. Higher health monitoring awareness was observed in patients with higher education.[13] Disease duration was measured to determine the patient's condition. In RA patients, the shortest disease duration was 3 months and the longest was 204 months, whereas in OA patients, the shortest disease duration was 5 months and the longest disease duration was 300 months (Fig. 1). The disease duration range was very wide with various conditions.
|
Table 2: Drug therapy of RA and OA patients |
||
|
Group |
Drug therapy |
Number of patients |
|
RA patient (n = 30) |
Methotrexate |
18 |
|
Methotrexate, chloroquine |
4 |
|
|
Methotrexate, sulfasalazine |
2 |
|
|
Methotrexate, chloroquine, sulfasalazine |
2 |
|
|
Cyclosporine |
2 |
|
|
Without DMARD |
2 |
|
|
OA patient (n =18) |
Glucosamine Glucosamine, calcium carbonate Glucosamine, chondroitine Glucosamine, calcium carbonate, chondroitine Eperisone hydrochloride Meloxicam Analgesic |
5 1 2 3 3 2 2 |
RA patients receive combination therapies, i.e. disease-modifying antirheumatic drugs (DMARDs), non-steroidal anti-inflammatory drugs (NSAIDs), corticosteroid, and analgesic. RA patients (93.3%) received a combination of DMARDs with folic acid, methylprednisolone, and calcium carbonate (Table 2). Methotrexate is a folic acid antagonist, so it must be combined with folic acid to reduce its side effects.[14,15] Methylprednisolone is used to control pain and synovitis. The long-term effect of corticosteroids causes osteoporosis, so it must be combined with vitamin D and calcium carbonate to minimize bone loss.[16]
86.7% of RA patients received methotrexate as DMARD because it was the first choice of DMARD to RA treatment. Methotrexate mechanism is an inhibitor of cytokine production and purine biosynthesis, and stimulator of adenosine release.[17] The interviews showed that methotrexate was used in RA patients with disease duration ranging from 9 to 204 months. A combination of two or three DMARDs was given to 26.7% of RA patients, due to its higher level of effectiveness than a single DMARD.[18] Cyclosporine was given to 6.7% of RA patients, because of the methotrexate side effects it causes, namely nausea and diarrhea. DMARD was not administered to the first attending RA patients (6.7%); they were instead given an analgesic and were provided with clinical testing to support the AR diagnosis. Analgesics, such as paracetamol, ibuprofen or diclofenac sodium, were given to 33.3% of RA patients to reduce pain due to joint inflammation. Glucosamine was also given to 6.7% of RA patients to build new cartilage structure. Sulfated glucosamine has many negative charges that would inhibit the function of inflammatory cells.[19]
The knee OA patients received combined therapies, such as glucosamine, chondroitin, and calcium carbonate (Table 2). A single or a combination of glucosamine was given to 61.1% of knee OA patients, because of reduced cartilage. Knee OA patients required additional glucosamine to increase the formation of new cartilage.[19] Chondroitin sulfate was given to 27.78% of knee OA patients because it stimulates proteoglycan production and prevents cartilage degradation.[20] The knee OA patients were also given calcium carbonate to prevent bone loss,[16] because of increased risk of bone loss and fracture.[21] Eperisone hydrochloride was used for disease with muscle stiffness and pain.[22] Meloxicam, a COX-2 preferential, was given as an anti-inflammatory and analgesic with fewer GI adverse effects.[23]
Determination of serum COMP levels
Cartilage oligomeric matrix protein (COMP) is a non-collagenous biomarker that is currently under investigation.[24] The serum COMP levels showed an outlier, i.e. two for RA patients, one for knee OA patients, and four for normal individuals. Thus, they were excluded from calculations. The Shapiro-Wilk normality test showed normally distributed data for RA patients (W=0.947 and p=0.17), knee OA patients (W=0.966 and p=0.75), and normal individuals (W=0.962 and p= 0.44) (Fig. 2).
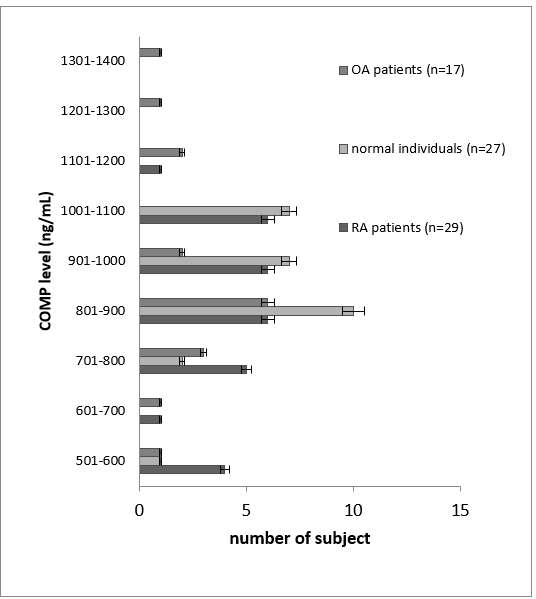
Figure 2. Distribution of serum COMP levels. The serum COMP levels of normal individuals were higher than RA and OA patients.
The serum COMP levels of RA patients, knee OA patients, and normal individuals were 873.16 ± 165.6, 911.43 ± 227.34, and 927.26 ± 90.38 ng/mL, respectively. Statistical analysis showed no significant difference in serum COMP levels (p=0.46) among the groups. The serum COMP levels of RA and OA patients were lower than normal individuals due to patient adherence in taking prescribed drugs (Fig. 3), so that it maintains the life quality and stabilizes patients’ disease activity.[25]

Figure 3. Distribution of serum COMP levels to disease duration. Disease duration did not affect serum COMP levels.
Patient adherence in taking prescribed drugs supported the statistical analysis that showed no correlation between serum COMP levels and disease duration in RA patients (p=0.52, rho=0.20) and the knee OA patients (p=0.65, rho=0.25). It was suggested as a successful treatment because it can maintain serum COMP levels. It was evident that the disease duration did not affect serum COMP levels, as long as the patient obediently consumes the prescribed drugs. Patient adherence was essential for controlling chronic diseases, such as RA and knee OA. The goal of proper and regular therapy is to maintain the quality of life and suppress disease activity.[26] All patients felt the pain if they did not take the drug immediately. This experience makes the patient adherent to take the prescribed drugs. These results were consistent with the previous studies in RA patients at Purwakarta District, West Java, Indonesia.[27]
Conclusions
Drug treatment maintains the serum COMP levels of RA and the knee OA patients.
Conflicts of interest
There is no conflict of interest.
References
- Heinegard D, Saxne T. The role of the cartilage matrix in osteoarthritis. Nat Rev Rheumatol, 2011; 7: 50-56.
- Saxne T, Mansson B, Heinegard D. New frontiers in pathogenesis and treatment, Rheumatoid Arthritis. Firestein G, Panayi G, Wollheim F, eds. Oxford, UK: Oxford University Press; 2006.
- Heinegard D. Proteoglycans and more-from molecules to biology. Int J Exp Pathol, 2009; 90: 575-86.
- Heathfield TF, Onnerfjord P, Dahlberg L, Heinegard D. Cleavage of fibromodulin in cartilage explants involves removal of the N-terminal tyrosine sulfate-rich region by proteolysis at a site that is sensitive to matrix metalloproteinase-13. J Biol Chem, 2004; 279: 6286-6295.
- Kraus VB, Burnett B, Coindreau J, Cottrell S, Eyre D, Gendreau M, Garginer J, Garnero P, Hardin J, Henrotin Y, Heinegard D, Ko A, Lohmander S, Matthews G, Menetski J, Moskowitz R, Persiani S, Poole R, Rousseau JC, Todman M. Application of biomarkers in the development of drugs intended for the treatment of osteoarthritis. Osteoarthritis Cartilage, 2011; 19: 515-542.
- Saxne T, Heinegard D. Cartilage oligomeric matrix protein: a novel marker of cartilage turnover detectable in synovial fluid and blood. Br J Rheumatol, 1992; 31: 583-591.
- Halasz K, Kassner A, Morgelin M, Heinegard D. COMP acts as a catalyst in collagen fibrillogenesis. J Biol Chem, 2007; 282: 31166-31173.
- Thur J, Rosenberg K, Nitsche DP, Pihlajamaa T, Ala-Kokko L, Heinegard D, Paulsson M, Maurer P. Mutations in cartilage oligomeric matrix protein causing pseudoachondroplasia and multiple epiphyseal dysplasia affect binding of calcium and collagen I, II, and IX. J Biol Chem, 2001; 276: 6083-6092.
- Neidhart M, Hauser N, Paulsson M, Dicesare PE, Michel BA, Haulselmann HJ. Small fragments of cartilage oligomeric matrix protein in synovial fluid and serum as markers for cartilage degradation. Br J Rheumatol, 1997; 36: 1151-1160.
- Sharif M, Saxne T, Shepstone L. Relationship between serum cartilage oligomeric matrix protein levels and disease progression in osteoarthritis of the knee joint. Br J Rheumatol, 1995; 34: 306-3
- Williams FM, Spector TD. Biomarkers in osteoarthritis. Arthritis Res Ther, 2008; 10: 101-109.
- Saptarini NM, Pratiwi DE, Febrina E, Wibowo MS, Gusdinar T. Correlation Study Of Disease Activity Score And Serum Cartilage Oligomeric Matrix Protein Level Of Rheumatoid Arthritis Patients In Bandung, Indonesia. Asian J Pharm Clin Res, 2017; 10 (1): 290-293.
- Saptarini NM, Wibowo MS, Gusdinar T. Correlation Study of Age, Disease Duration, dan Erythrocyte Sedimentation Rate Among The Indonesian Rheumatoid Arthritis Patients. Inter J Pharm Pharm Sci, 2015; 7 (11): 274-277.
- American College of Rheumatology Subcommittee on Rheumatoid Arthritis. Guidelines for the management of rheumatoid arthritis: 2002 Update. Arthritis Rheum, 2002; 46 (2): 328-346.
- Borchers AT, Keen CL, Cheema GS, Gershwin ME. The use of methotrexate in rheumatoid arthritis. Seminar of Arthritis Rheum, 2004; 34 (1): 465-483.
- McIlwain HH. Glucocorticoid-induced osteoporosis: Pathogenesis, diagnosis, and management. Prev Med, 2003; 36 (2): 243-249.
- Pincus T, Ferraccioli G, Sokka T, Larsen A, Rau R, Kushner I, Wolfe F. Evidence from clinical trials dan long-term observational studies that disease-modifying anti-rheumatic drugs slow radiographic progression in rheumatoid arthritis: Updating a 1983 review. Rheumatology, 2002; 41 (12): 1346-1356.
- Goldbach-Mansky R, Lipsky PE. New concepts in the treatment of rheumatoid arthritis. Annual Rev Med, 2003; 54: 197-216.
- Theoharides TC, Patra P, Boucher W, Letourneau R, Kempuraj D, Chiang G, Jeudy S, Hesse L, Athanasiou A. Chondroitin sulfate inhibits connective tissue mast cells,” Br J Pharmacol, 2000; 131: 1013-10
- Bucsi L, Poór G. Efficacy and tolerability of oral chondroitin sulfate as a symptomatic slow-acting drug for osteoarthritis (SYSADOA) in the treatment of knee osteoarthritis. Osteoarthritis Cartilage, 1998; 6: 31-36 (suppl A).
- Esdaile JM, Goicochea M, Lacaille D. Comorbidity in rheumatoid arthritis, Rheumatoid arthritis. Hochberg MC, Silman AJ, Smolen JS, et al., eds., Philadelphia: Mosby Elsevier; 2009.
- Murayama KK, Atsumi TT, Ajika EN, Akamura T. Clinical application of eperisone hydrochloride to neurogenic bladder. Hinyokika Kiyo. 1984; 30: 403-408.
- Ahmed M, Khanna D, Furst DE. Meloxicam in rheumatoid arthritis. Expert Opinion on Drug Metabolism and Toxicology, 2005; 1 (4): 739-751.
- Lindqvist E, Eberhardt K, Bendtzen K, Heinegard D, Saxne T. Prognostic laboratory markers of joint damage in rheumatoid arthritis. Annals of the rheumatic diseases, 2005; 64 (2): 196-201.
- Schuna AA. Rheumatoid arthritis, Pharmacotherapy: A Pathophysiologic Approach. 7 ed., Dipiro JT, Talbert RL, Yee GC, et al., eds., Singapore: Mc Graw Hill Medical; 2007.
- Ibrahim O, Jirjees F, Mahdi H. Barriers affecting compliance of patients with chronic diseases: a preliminary study in united arab emirates (UAE) population. Asian J Pharm Clin Res, 2011; 4: 42-45.
- Saptarini NM, Wibowo MS, Gusdinar T. Erythrocyte sedimentation rate as an indicator of compliance of rheumatoid arthritis patients: a case study in West Java, Indonesia. Mahidol Univ J Pharm Sci. 2016; 43 (2): 55-62.
Contact SPER Publications
SPER Publications and
Solutions Pvt. Ltd.
HD - 236,
Near The Shri Ram Millenium School,
Sector 135,
Noida-Greater Noida Expressway,
Noida-201301 [Delhi-NCR] India
