How to cope with pregnancy of unknown location: construction of a scoring system
Marjan Haji Heshmati1, Maliheh Arab2*, Robabeh Ghodsi-Ghassemabadi3
1 Assistant of Obstetrics and Gynecology, Shahid Beheshti University of Medical Sciences, Tehran, Iran. 2 Professor of Gynecology-Oncology, Shahid Beheshti University of Medical Sciences, Tehran, Iran. 3 Ph.D. Candidate, Department of Biostatistics, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran.
Correspondence: Maliheh Arab. Professor of Gynecology-Oncology, Shahid Beheshti University of Medical Sciences, Tehran, Iran. Email: drmarab @ yahoo.com.
|
ABSTRACT Background: Women presenting with abdominal pain in their first trimester of pregnancy could have EP, abortion or normal intrauterine pregnancy. The decision about PUL (Pregnancy of unknown Location) is often difficult because of serious medical complications or the moral consequences of misdiagnosis. Objective: Construct a scoring system to triage EP among PUL patients in pregnant women with abdominal pain. Method: Medical history, symptoms and signs, laboratory data, and sonography of 522 pregnant women presented with abdominal pain in their first trimester at Imam Hossein Medical Center in Tehran were recorded through a questionnaire. Patients were compared in EP and non-EP groups (abortion and normal pregnancy). Based on the univariate analysis, statistically significant variables were used in the construction of a logistic regression model, and finally a clinical scoring system was developed. Results: In this cross-sectional study, medical record of 522 cases including 183 (36%) EP and 334 (64%) Non-Ep patients were reviewed. Among 92 studied variables, 41 items were significantly correlated with EP diagnosis (P-Value <0.05). Six variables (duration of pain before hospitalization less than 9 hours, gestational age less than 65 days, leukocyte over 8600 /ml, abdominal tenderness (superficial, deep or generalized), abdominal or pelvic free fluid, and pain (intermittent, persistent, obscure, shifting, and radiating)), were identified as predictors of EP detection in the logistic regression model and scoring system. Based on the proposed scoring system, patients with scores 0 to 4 were considered as low-risk (5% probability), scores 5-8 as moderate-risk (63% probability), and scores 9 to 14 were considered as high-risk (95% probability) for EP.Conclusions: The present scoring system could help clinicians to find EP among PUL patients. In a score of more than 9 (95% probability), interventions (laparoscopy or methotrexate) might be considered. Keywords: pregnancy of unknown location, ectopic pregnancy, abdominal pain, scoring |
Introduction
Women presenting with vaginal bleeding or pain in the first trimester of pregnancy may have ectopic pregnancy (EP), abortion, or normal intrauterine pregnancy [1, 2]. The term pregnancy of unknown location (PUL) refers to women with positive HCG whom sonography does not show any gestational sac in the uterus or any extra uterine site [3]. The decision about PUL is often difficult because of serious medical complications or the moral consequences of misdiagnosis. Inaccurate interventions may cause termination of a normal pregnancy, while an untreated EP may lead to tubal rupture, shock, or even death [4].
PUL is not a final diagnosis. It is a transit state, and patient will be diagnosed finally with EP, abortion or normal intrauterine pregnancy. Since EP causes mortality, morbidity, and infertility, and its promoted diagnosis is a key to prevent fatal consequences [1].
Ectopic pregnancy usually causes pain in most and even all patients. When Tilt Test is positive, and the patient is in shock, there is no doubt in performing the surgical procedure [4]. However, in patients whose HCG test still does not meet the cut-off titer of 1000-2000 for detection of the pregnancy sac in the ultrasound [5, 6], there is an ambiguity in the diagnosis and treatment.
Variables considered in the medical literature to be associated with ectopic pregnancy have been the history of addiction, abortion, multiple partners, contraception methods, infertility, history of ectopic pregnancy, medical disease, history of surgery, pelvic infection, abnormal bleeding, pain, nausea and vomiting, age, history of pregnancy, vital signs, hemoglobin, leukocyte count, HCG titre, and sonographic findings [7]. The aim of the present study was to construct a scoring system based on clinical and paraclinical tests to guide clinicians for the management of PUL presenting with pain.
Materials and Methods
In this cross-sectional study, clinical symptoms, sings, laboratory tests, and sonographic findings of women with positive HCG in first trimester and lower abdominal pain presenting to Imam Hossein Medical Center located in Tehran, Iran were recorded through a questionnaire. Women were categorized into two groups of Ep and Non-EP. Women whose medical record were incomplete, were excluded from this study. The data set of questionnaires included 64 questions and 92 variables. The data of cases in two groups of EP and Non-Ep were compared. Significant meaningful variables regarding EP outcome were entered for the construction of the logistic regression model and the scoring system.
Study variables:
EP was the outcome of the study. Various factors associated with EP included risk factors, clinical signs, physical examination and paraclinical tests. Variables included in the questionnaire were as follow: the history of addiction, abortion, multiple partners, contraception methods, infertility, history of ectopic pregnancy, medical disease, history of surgery, pelvic infection, abnormal bleeding, pain, nausea and vomiting, age, history of pregnancy, vital signs, hemoglobin, leukocyte count and HCG titre.
Statistical analysis:
Categorical data are represented as frequency and percent, and quantitative data are presented as mean and standard deviation or median and IQR. Independent sample t-test, Mann-Witheny test, and chi-square test were used to compare characteristics between the two groups. Statistically significant variables in univariate analysis with frequency more than 10 percent were included in the multiple logistic regressions using stepwise backward elimination, with EP as the dependent variable. The continuous independent variables were categorized based on Youden index cut off point for inclusion into the model. Coefficients of each significant variables in the final model were used to develop a risk score. To construct the score of each significant variable, the coefficient of each variable was divided by the lowest β value in the model and rounded to the nearest integer. The final value of risk score predictive model was the sum of the risk scores mentioned above. Hosmer-Lemeshow goodness of fit test was used to evaluate how well the model was calibrated.The area under the receiver operation characteristic (ROC) curve was calculated to evaluate the accuracy of the prediction model.
Results:
Of 574 cases investigated, 52 were excluded from the study due to the deficiencies in data recording, and finally 522 cases were included in the study. 188 patients (36%) were in the EP group, and 334 (64%) were in the non-EP group. Among 92 studied variables, 36 variables were significantly correlated with EP diagnosis (Table 1). The results of backward stepwise logistic regression analysis identified 6 significant variables, including duration of pain before hospitalization less than 9 hours, gestational age less than 65 days, leukocyte over 8600 /ml, superficial, deep and generalized tenderness, abdominal or pelvic free fluid, intermittent, persistent, obscure, shifting, or radiating pain. The results of logistic regression has been represented in Table 2.
|
Table 1. Comparison of qualititative variables in EP and Non-EP groups |
|||||
|
Variable |
Category |
EP |
Non-EP |
P value |
|
|
frequency (percent) |
frequency (percent) |
||||
|
Contraception |
Non |
97 (51.6%) |
165 (49.4%) |
||
|
|
IUD or TL Use |
18 (9.6%) |
8 (2.4%) |
0.001 |
|
|
|
Other methods* |
73 (38.8%) |
161 (48.2%) |
||
|
Infertility |
|
25 (13.3%) |
19 (5.7%) |
0.003 |
|
|
Adhesiolysis |
|
4 (2.1%) |
0 (0%) |
0.016 |
|
|
Pain Start |
Gradually |
45 (23.9) |
170 (50.9) |
<0.001 |
|
|
|
Sudden or post-coital |
143 (76.1) |
164 (49.1%) |
|
|
|
Pain Quality |
Continuous pain |
98 (52.1%) |
16 (4.8%) |
<0.001 |
|
|
|
Intermittent pain |
44 (23.4%) |
260 (77.8%) |
|
|
|
|
Radiating pain |
15 (8%) |
4 (1.2%) |
|
|
|
|
Shifting or vague pain |
31 (16.5%) |
54 (16.2%) |
||
|
Pain fluctuation |
Increasing pain |
142 (75.5%) |
88 (26.3%) |
<0.001 |
|
|
|
Decreasing pain |
24 (12.8%) |
104 (31.1%) |
|
|
|
|
Fluctuating pain |
22 (11.7%) |
142 (42.5%) |
|
|
|
Primary pain |
One-sided pain |
153 (95.6%) |
7 (4.4%) |
<0.001 |
|
|
|
Two-sided pain |
35 (9.7%) |
327 (90.3%) |
|
|
|
Nausea and vomiting |
|
77 (41%) |
35 (10.5%) |
<0.001 |
|
|
Urinary symptoms |
|
15 (8%) |
11 (3.3%) |
0.018 |
|
|
Tilt sign |
|
43 (22.9%) |
8 (2.4%) |
<0.001 |
|
|
Tenderness |
no |
65 (34.6) |
317 (94.9) |
<0.001 |
|
|
|
Tenderness with deep palpation |
67 (35.6%) |
12 (3.6%) |
|
|
|
|
Tenderness with superficial palpation |
29 (15.4%) |
3 (0.9%) |
|
|
|
|
Generalized tenderness |
27 (14.4%) |
2 (0.6%) |
|
|
|
|
LLQ |
49 (39.8%) |
1 (5.9%) |
|
|
|
|
Two-sided |
16 (13%) |
7 (41.2%) |
|
|
|
Rebound tenderness |
|
52 (27.8%) |
2 (0.6%) |
<0.001 |
|
|
Cough sign |
|
2 (1.1%) |
2 (0.6%) |
<0.001 |
|
|
Mass palpation |
|
3 (1.6%) |
0 (0%) |
0.046 |
|
|
Normal sonography |
|
1 (0.6%) |
31 (9.3%) |
<0.001 |
|
|
Free fluid |
|
152 (77.6%) |
44 (22.4%) |
<0.001 |
|
|
Myoma |
|
14 (7.4) |
12 (3.6) |
0.052 |
|
|
Gestational age (days) |
|
55.62 |
68.21 |
<0.001 |
|
|
Pulse rate (/min) |
|
94.57±12.68 |
89.67±8.01 |
<0.001 |
|
|
Diastolic blood pressure(mm.Hg) |
|
64.41±17.29 |
71.13±8.73 |
<0.001 |
|
|
Pain duration (hours) |
|
5 (4) |
13 (15) |
<0.001 |
|
|
Hemoglobin 1 (g/dl) |
|
11.21±1.65 |
11.85±1.24 |
<0.001 |
|
|
Table 2. Variables associated with EP based on logistic regression result |
||||||||
|
|
|
|
|
|
|
|
95% CI for OR |
|
|
Variable |
category |
|
Beta |
S.E. |
P value |
OR |
Lower |
Upper |
|
Pain duration |
≤9 vs. >9 |
|
0.910 |
.337 |
0.007 |
2.49 |
1.284 |
4.806 |
|
LMP |
≤65 vs. >65 |
|
1.44 |
.381 |
<0.001 |
4.23 |
2.005 |
8.932 |
|
Leukocyte |
>8600 vs. ≤8600 |
|
0.732 |
.326 |
0.025 |
2.08 |
1.096 |
3.941 |
|
Tenderness |
no |
|
|
- |
|
|
|
|
|
Deep palpitation |
|
2.09 |
.452 |
<0.001 |
8.06 |
3.324 |
19.553 |
|
|
Superficial palpitation |
|
1.37 |
.786 |
0.081 |
3.94 |
.845 |
18.387 |
|
|
Generalized palpitation |
|
1.95 |
.891 |
0.028 |
7.05 |
1.229 |
40.423 |
|
|
Free fluid |
no |
|
|
- |
|
|
|
|
|
mild |
|
1.77 |
.348 |
<0.001 |
5.86 |
2.962 |
11.610 |
|
|
moderate or sever |
|
2.56 |
.599 |
<0.001 |
12.98 |
4.011 |
42.021 |
|
|
Pain quality |
intermittent |
|
|
- |
|
|
|
|
|
continuous |
|
2.13 |
.440 |
<0.001 |
8.42 |
3.554 |
19.929 |
|
|
vague or shifting |
|
0.950 |
.399 |
0.017 |
2.59 |
1.183 |
5.647 |
|
|
radiating |
|
1.99 |
.835 |
0.017 |
7.37 |
1.433 |
37.861 |
|
These variables as predictors of EP detection have been presented in the constructed model as below:
log(P1-p)=-4.61+0.910 Pain duration+1.44LMP+0.732Leukocyte+2.09 Deep palpation+1.95 Generalized palpation+1.37 Superficial palpation+1.77 mild free fluid+2.56 moderate|severe free fluid+2.13 continuous pain+0.950vague|shifting pain+1.99 radiating pain
The Hosmer-Lemeshow goodness of fit test (p=0.847) showed that there was no statistically significant difference between the predicted and observed outcomes. The area under the ROC curve of the constructed model was 0.953 with 81.8% sensitivity, and 94.3% specificity for EP diagnosis. To apply this mathematical equation for clinicians, a clinical scoring system was proposed. To determine the risk of EP, each coefficient of the prediction model was divided by the smallest coefficient (leukocyte coefficient). It should be noted that each of the meaningful variables (Table3) scored 1 to 4. Then, each patient could receive total scores from 0 to 14 based on this scoring system. EP probability increased significantly with increasing score of EP (P <0.001 for trend). The area under the curve (AUC) for the EP Risk Scoring system had good diagnostic accuracy (AUC: 0.952, 95% CI) as shown in Fig 1.

Figure 1. ROC Curve for Predicting EP based on Clinical Presentation, Laboratory Data, and Sonography (AUC= 0.952)
|
Table 3. Risk Score for EP prediction |
||
|
Clinical, sonography, and medical parameters |
Score |
|
|
Pain duration ≤ 9 hours |
1 |
|
|
Gestational age ≤ 65 days |
2 |
|
|
Leukocyte > 8600 /ml |
1 |
|
|
Tenderness |
Deep palpation |
3 |
|
Generalized palpation |
3 |
|
|
Superficial palpation |
2 |
|
|
Free fluid |
mild free fluid |
2 |
|
Moderate/severe free fluid |
4 |
|
|
Quality of pain |
Continuous pain |
3 |
|
vague or shifting pain |
1 |
|
|
Radiating pain |
3 |
|
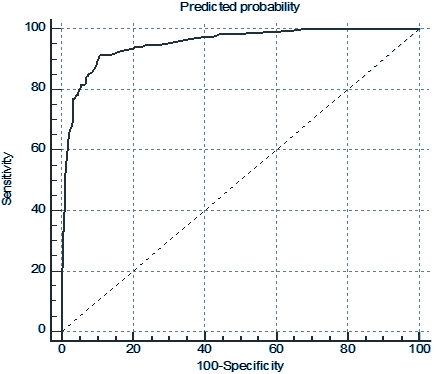
Figure 2. Probability of EP based on EP risk scoring system
Based on the proposed scoring system, patients with scores 0 to 4 were considered as low-risk(5% probability), scores 5-8 as moderate-risk (63% probability), and scores 9 to 14 were considered as high-risk (95% probability) for EP (Figure 2).
Discussion:
EP is one of the leading causes of death in the first trimester of pregnancy, so its correct and timely diagnosis can prevent mortality and morbidity [4]. Scoring system constructed in the present study differentiated moderate and high risk cases for EP with probability of 63% and 95%; respectively. Clinicians might decide to operate in high probability or observe in low probability cases.
Many studies have been conducted to help clinical management of PUL, specially in presentation of pain. In a study, Kurt. T Barnhart et al. [8], designed scoring for nonviable pregnancy (Ep or abortion) in PUL with bleeding and/or abdominal pain, the results revealed age, race, parity, history of abortion, history of spontaneous abortion, history of pelvic surgery, history of previous caesarean section, history of PID, history of Gonorrhea or chlamydia, Gonorrhea or Chlamydia were present at the time of entry, HCG levels, and pain in differentiation of the nonviable pregnancy.
In a systematic review by Bobdiwala et al. [5] for the diagnostic protocol for managing PUL, the M4 model was the best model to predict the EP's final outcome. This logistic regression model was based on the initial HCG and HCG Ratio (HCG 48 hours/ HCG 0 hours) for prediction of EP. In this study, HCG levels did not significantly correlate with EP probability, probably due to below 2000 HCG titer in all cases and nearly more complicated clinical condition that presented with pain resulting in surgery.
In a study by Camille Mimoun et al. [9], five items had a significant association with EP, duration of bleeding more than 24 hours (score = 25), cough sign (score = 15), one-sided pelvic pain (score = 15), brown discharge (score = 15), and no frequent need to change sanitary towels (score = 30).These five variables were scored, and the patients were divided into low-risk and high-risk group with score of <25 and > 70; respectively.
In a study by Banhart et al. [10], clinical data from women with PUL were analysed and an EP risk-score was determined. Some characteristics including age, race, parity, history of elective abortions, and history of miscarriage had a meaningful relationship with EP.
In the present study, based on the pain duration, gestational age, leukocytosis, abdominal tenderness, free fluid in sonography, and quality of pain, patients’ probability of EP diagnosis was revealed. In high-risk group (95% probability), laparoscopic intervention, and in the moderate-risk group (63 % probability), close observation might be considered. Pain and abdominal tenderness if present, accepted 6 scores at least moderate risk with 63% probability, which is consistent with Camille Mimoun et al. study [9].
Results of the present study just can be conducted in PUL with pain cases. Validation of the present scoring system in separate population of patients with PUL -with or without pain- has been suggested.
Limitations
In this study, patients with PUL suffering from abdominal pain were examined, and painless PULs were excluded. As expected, the reason for this was the referral of more patients with abdominal pain to the emergency department. It is recommended that a study be conducted in the future on the examination of all PUL cases (with or without abdominal pain).
Conclusion
In conclusion, in patients with abdominal pain and positive HCG test, whose ultrasound was equivocal and HCG titer did not reach the discriminative zone, the scoring system of the present study can find high-risk patients with 95% probability of EP, candidate of intervention with laparoscopy and/or methotrexate therapy.
References
- Barnhart K, van Mello NM, Bourne T, Kirk E, Van Calster B, Bottomley C, Chung K, Condous G, Goldstein S, Hajenius PJ, Mol BW. Pregnancy of unknown location: a consensus statement of nomenclature, definitions, and outcome. Fertility and sterility. 2011 Mar 1; 95 (3):857-66.
- Barnhart KT, Sammel MD, Gracia CR, Chittams J, Hummel AC, Shaunik A. Risk factors for ectopic pregnancy in women with symptomatic first-trimester pregnancies. Fertility and sterility. 2006 Jul 1; 86 (1):36-43.
- Bobdiwala S, Al-Memar M, Farren J, Bourne T. Factors to consider in pregnancy of unknown location. Women's Health. 2017 Aug; 13(2):27-3
- Loss Ep. ACOG Practice Bulletin.
- Bobdiwala S, Saso S, Verbakel JY, Al‐Memar M, Van Calster B, Timmerman D, Bourne T. Diagnostic protocols for the management of pregnancy of unknown location: a systematic review and meta‐analysis. BJOG: An International Journal of Obstetrics & Gynaecology. 2019 Jan; 126 (2):190-8.
- Condous G, Kirk E, Lu C, Van Huffel S, Gevaert O, De Moor B, De Smet F, Timmerman D, Bourne T. Diagnostic accuracy of varying discriminatory zones for the prediction of ectopic pregnancy in women with a pregnancy of unknown location. Ultrasound in Obstetrics and Gynecology: The Official Journal of the International Society of Ultrasound in Obstetrics and Gynecology. 2005 Dec; 26 (7):770-5.
- Li C, Zhao WH, Zhu Q, et al. Risk factors for ectopic pregnancy: a multi-center case-control study. BMC Pregnancy Childbirth 2015; 15:18
- Barnhart KT, Sammel MD, Takacs P, Chung K, Morse CB, O'Brien KO, Allen-Taylor L, Shaunik A. Validation of a clinical risk scoring system, based solely on clinical presentation, for the management of pregnancy of unknown location. Fertility and sterility. 2013 Jan 1; 99(1):193-
- Mimoun C, Fauconnier A, Varas C, Huchon C. Is a Self-Assessed Questionnaire Useful for the Diagnosis of Ectopic Pregnancy in Hospitalized Patients?. PloS one. 2016 Nov 10; 11(11):e0155054.
- Barnhart KT, Casanova BC, Sammel MD, Timbers K, Chung K, Kulp JL. Prediction of location of a symptomatic early gestation based solely on clinical presentation. Obstetrics and gynecology. 2008 Dec; 112 (6):1319.
Contact SPER Publications
SPER Publications and
Solutions Pvt. Ltd.
HD - 236,
Near The Shri Ram Millenium School,
Sector 135,
Noida-Greater Noida Expressway,
Noida-201301 [Delhi-NCR] India
